Docket #: S12-089
Customizable, Porous Tissue Engineering Scaffold for 3D Cell Proliferation
Hydrogel-based tissue engineering scaffolds are widely used for culturing cells in three dimensions (3D) due to their tissue-like water content, tunable biochemical and physical properties, and ease of cell encapsulation and distribution in 3D. However, most conventional hydrogels lack the macroporosity desirable for efficient cell proliferation and migration, and have limited flexibility when subject to mechanical load. Microfiber-based scaffolds, on the other hand, are inherently macroporous and flexible, but are often associated with cell-unfriendly fabrication procedures which make microfibers unsuitable for direct cell-encapsulation.
To overcome these limitations, Stanford researchers have created a novel scaffold material consisting of crosslinkable, microribbon-like elastomers, which can assemble into highly macroporous scaffolds that facilitate cell encapsulation and support cell proliferation in 3D.

The biochemical composition, macroporosity level, and mechanical properties of the microribbon-based scaffolds can be independently adjusted to influence cell behaviors associated with biochemical, topographical and mechanical cues. Furthermore, the geometry of microribbons enables the microribbon-based scaffolds to sustain large deformation and mechanical stress.
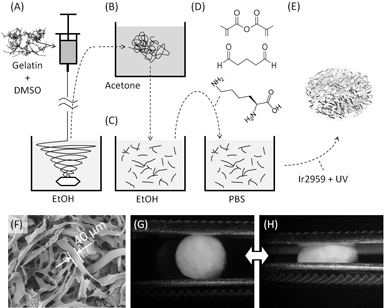
In published results, the researchers showed that, when encapsulated in the microribbon-based scaffold, human adipose derived stromal cells proliferated up to 30-fold within 3 weeks. Furthermore, microribbon-based scaffolds demonstrate great flexibility and can sustain up to 90% strain and 3 MPa stress without failing. Such unique macroporosity and flexibility of the microribbon-based scaffolds make them promising for engineering shock-absorbing tissues such as cartilage and intervertebral discs.
Applications
- 3D scaffolds of customized macroporosity, biochemical properties, and mechanical properties
- Implants customized for specific tissue-regeneration (ex: skin, cartilages, bones)
- A novel research platform for studying cell-responses to different microenvironments of customized biochemical, biophysical, and topographical cues
- Substrates for 3D cell culture
Advantages
- Highly Tunable Properties – control over porosity, biochemical and mechanical properties
- Direct Cell-Encapsulation – enables control over cell distribution
- Flexibility – can sustain up to 90% strain and return to its original shape
- This technology enables combination of the benefits of both hydrogel and microfiber-based tissue engineering scaffolds
- Flexible
- Macroporous
- Direct Cell-Encapsulation
Publications
- Li-Hsin Han et al. "Microribbon-Like Elastomers for Fabricating Macroporous and Highly Flexible Scaffolds that Support Cell Proliferation in 3D". Sep 2012. Adv Funct Mat. 10.1002/adfm.201201212
- Han LH, Chung MT, Deveza L, Jiang X, Conrad B, Wang A, Butte MJ, Longaker MT, Wan D, Yang F. Microribbon-based hydrogels accelerate stem cell-based bone regeneration in a mouse critical-size cranial defect model J Biomed Mater Res A. 2016 Jun;104(6):1321-31.
- Gegg C, Yang F. Spatially patterned microribbon-based hydrogels induce zonally-organized cartilage regeneration by stem cells in 3D Acta Biomater 2020 Jan 1;101:196-205. Epub 2019 Oct 19. PMID: 31634627.
- Ueno M, Lo CW, Barati D, Conrad B, Lin TH, Kohno Y, Utsunomiya T, Zhang N, Maruyama M, Rhee C, Romero-Lopez M, Khan T, Tong X, Yao Z, Zwingenberger S, Yang F, Goodman SB IL-4 Overexpressing Mesenchymal Stem Cells within Gelatin-Based Microribbon Hydrogels Enhance Bone Healing in a Murine Long Bone Critical-size Defect Model Journal of Biomedical Materials Research: Part A. 2020 May 3. Online ahead of print.
- Barati D, Gegg C, Yang F. Nanoparticle-mediated TGF-? Release from Microribbon-based Hydrogels Accelerates Stem Cell-based Cartilage Formation in vivo Annals of Biomedical Engineering 2020 Jul;48(7):1971-1981. Epub 2020 May 6.
- Tang Y, Tong X, Conrad B, Yang F Injectable and in situ Crosslinkable Gelatin Microribbon Hydrogels for Stem Cell Delivery and Bone Regeneration in vivo Theranostics 2020 May 15;10(13):6035-6047.
Patents
- Published Application: 20140017284
- Published Application: 20160331508
- Issued: 9,402,710 (USA)
- Issued: 10,004,587 (USA)
Similar Technologies
-
Hetero-assembling, tunable, and injectable hydrogels for cell encapsulation S08-065AHetero-assembling, tunable, and injectable hydrogels for cell encapsulation
-
Crosslinked chitosan-lactide matrices for the controlled delivery of therapeutic agents S13-081Crosslinked chitosan-lactide matrices for the controlled delivery of therapeutic agents
-
Drug-imprinted hydrogel for controlled-release wound healing therapy with FAK inhibitors S14-447Drug-imprinted hydrogel for controlled-release wound healing therapy with FAK inhibitors