Docket #: S18-314
Electrolysis Cell for Generating Concentrated Liquid and Gas Product Streams from CO or CO2
Stanford researchers in the Kanan group have developed a electrolysis cell for generating and extracting liquid and gas product streams from CO and CO2. Previous designs for electrosynthesis of C2+ products from CO or CO2 suffer from dilute product streams due to low reactant concentration and mixed phase products. Reduction of CO to C2+ products complicates matters by producing mixed phase products: gaseous ethylene or liquid products (acetic acid, ethanol, propanol etc). To overcome this challenge, this cell utilizes interdigitated flow fields, combined with gas diffusion electrodes (GDEs) and a Nafion membrane to simultaneously attain high current density, high selectivity and high single pass conversion (68%) of CO reduction at moderate cell voltages (100 mA cm-2). Specifically, the Nafion membrane transports concentrated liquid products away from the GDE and is easily isolated from product gas streams (as high as 1.1M sodium acetate). Overall this design improves efficiency of water/ion flux to an electrode to extract concentrated product streams with potential for scaled up synthesis and expansion to CO2 electrolysis.
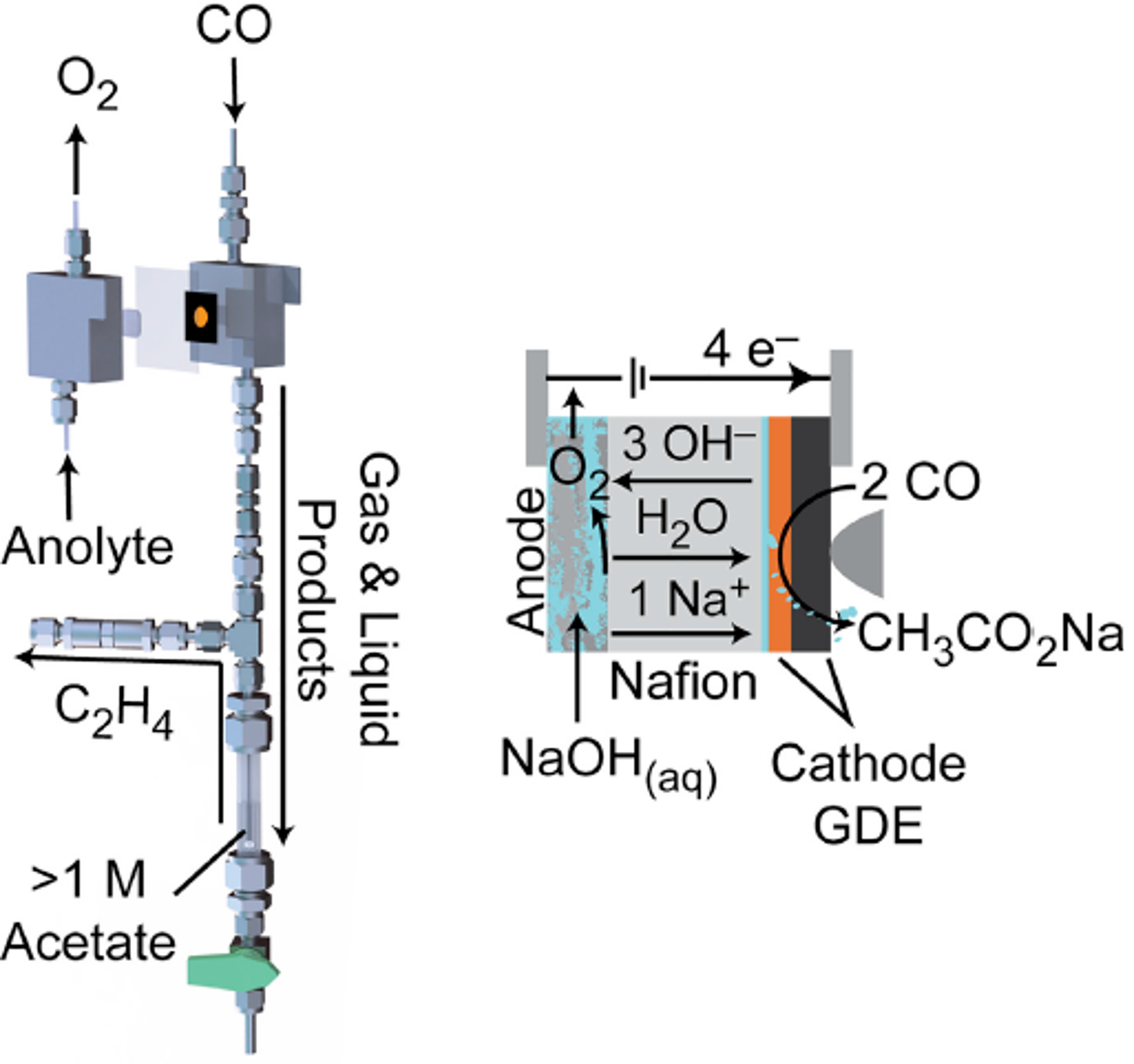
Photo description: Cell configuration and schematic of charge flow. Photo credit: Ripatti et al. Joule (2018).
Stage of Research
Applications
- Chemical synthesis of isotopically-labelled compounds
- Feedstock synthesis for hostile or resource constrained environments: potassium acetate as a growth medium
- Carbon sequestration and conversion: CO and CO2
Advantages
- Conversion of CO to C2+ products at high rates ((~849 µmol C2+ products cm–2 h–1)
- High single pass conversion (68%) and faradic efficiency (75%) for CO reduction at 100 mA cm-2
Publications
- Ripatti, D. S., Velman, T. R., and Kanan, M.W. "Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion." Joule 3.1 (2019): 240-256.
Related Links
Patents
- Issued: 11,479,871 (USA)
Similar Technologies
-
Enzymatic Detoxification of Aberrant NAD(P)H Tautomers for Improved Biosynthesis of Proteins and Biochemical Commodities S21-271Enzymatic Detoxification of Aberrant NAD(P)H Tautomers for Improved Biosynthesis of Proteins and Biochemical Commodities
-
Biological Production of Novel, High-Performance Polyesters S21-222Biological Production of Novel, High-Performance Polyesters
-
Biocatalytic production of para-hydroxybenzoic acid (p-HBA) from methanol and methane S20-336Biocatalytic production of para-hydroxybenzoic acid (p-HBA) from methanol and methane