Docket #: S13-022
Non-Invasive Imaging for the Detection of Human Embryonic Aneuploidy at the Blastocyst Stage
Stanford researchers have developed a non-invasive method of assessing chromosomal composition in human embryos at the blastocyst stage, thus potentially improving chances of success following in vitro fertilization (IVF). Experiments suggest that euploid human blastocysts exhibit cell cycle parameter timing that is different from aneuploid blastocysts and serve as the first example of the use of non-invasive imaging to predict ploidy at the blastocyst stage.
Given the high rate of human embryonic aneuploidy, this technology could improve IVF outcomes by reducing the transfer of aneuploid embryos, thus reducing the risk of high-order multiple pregnancies.
Figure
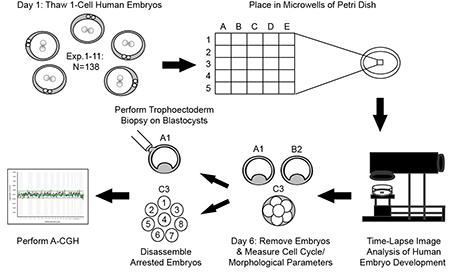
Stage of Research
Applications
- Human embryo transfer selection for IVF - distinguishes between euploid and aneuploid human blastocysts via non-invasive analysis of cell cycle parameter timing
Advantages
- Can lead to improved IVF success rate by improving embryo selection
- Non-invasive imaging method - Currently, the use of pre-implantation genetic screening (PGS) requires removal of the embryo to perform a biopsy.
- Improved prediction of embryo viability - fewer embryos could be transferred, thus reducing the risk of high-order multiple pregnancies
- Embryos can be selected sooner for implantation, reducing risk of epigenetic changes
Publications
- Friedman, B. E., Chavez, S. L., Behr, B., Lathi, R. B., Baker, V. L., & Reijo Pera, R. A. Non-invasive imaging for the detection of human embryonic aneuploidy at the blastocyst stage. Fertility and Sterility 98, no. 3 (2012): S38
- US Patent Application US20140349334 A1
Patents
- Published Application: 20140349334
- Issued: 9,404,908 (USA)
Similar Technologies
-
Predicting and Improving Embryo and Oocyte Viability Through Mechanical Measurements and Biomarkers S12-486Predicting and Improving Embryo and Oocyte Viability Through Mechanical Measurements and Biomarkers
-
Microfluidic guillotine for splitting cellular structures S18-227Microfluidic guillotine for splitting cellular structures
-
easyBAT: Simplified BAT Offering Rapid, Accurate, Automated and Accessible Solution for Food Allergy Diagnosis at the Point-of-Care S24-016easyBAT: Simplified BAT Offering Rapid, Accurate, Automated and Accessible Solution for Food Allergy Diagnosis at the Point-of-Care