Docket #: S21-108
REULR: Receptor Elimination by Ubiquitin Ligase Recruitment
Stanford researchers have developed REULR (Receptor Elimination by E3 Ubiquitin Ligase Recruitment), a mix and match nanobody (VHH)-based extracellular targeted protein degradation (eTPD) platform.
The toolbox is based on the development of high affinity, human and mouse cross-reactive nanobodies against five transmembrane PA-TM-RING-type E3 ubiquitin ligases: RNF128, RNF130, RNF167, RNF43, and ZNRF3 (Figure 1A). REULR molecules are heterobifunctional modalities than can enforce transmembrane E3 ligase interactions with a variety of disease-relevant target receptors (EGFR, EPOR, and PD-1) by induced proximity, resulting in effective degradation of the target receptor at varying levels. Unlike traditional antagonists that merely block protein function and rely on sustained high-occupancy, REULR offers catalytic, sustainable action with lower doses for extracellular/membrane targets (Figure 1B; left). In addition, the researchers designed E3 ligase self-degrading molecules, "Fratricide" REULRs (RNF128, RNF130, RENF167, RNF43, and ZNRF3), that allow downregulation of one or several E3 ligases from the cell surface (Figure 1B; right). Cell surface receptor modulation by REULR molecules potentially creates "tunable" therapeutic effects rather than all-or-none outcomes.
Figure
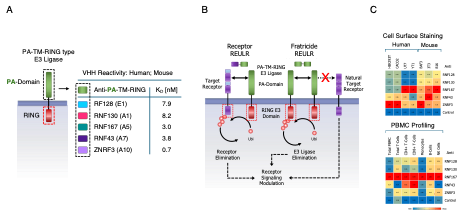
In addition to the therapeutic application of REULR molecules as an extracellular protein degradation (eTPD) platform, the monomeric REULR VHH binding modalities can be employed as human and mouse cross-reactive cell surface staining reagents (Figure 1C).The monomeric REULR nanobodies have affinities into the picomolar range, and demonstrate strong activity in cell surface staining (Flow), affinity capture and live cell applications (Incucyte). The 10-fold smaller size of nanobodies compared to traditional full length IgG provides for faster diffusion and are potentially more efficient staining of tissue sections or whole-mount samples. The smaller nanobody size also makes it an attractive tool for super-resolution microscopy, with the significantly closer proximity of the covalently conjugated fluorophore allowing more precise localization of the target antigen.
Stage of Development
In Vivo Research
Applications
- Binding reagents for research of cell surface proteins
- Therapeutic applications involving modulating activity of cell surface proteins
Advantages
- Allows modulation rather than all-or-none antagonism
- Potential for selective, tissue-specific applications
- Versatile and modular design - target one or multiple cell surface proteins
- Human and mouse cross-reactive VHHs
- High affinity, small size
Publications
- Siepe, D. H., Picton, L. K., & Garcia, K. C. (2023). Receptor elimination by E3 ubiquitin ligase recruitment (REULR): a targeted protein degradation toolbox. ACS synthetic biology, 12(4), 1081-1093.
Related Links
Patents
- Published Application: WO2022246130
- Published Application: 20240239891
Similar Technologies
-
A Candidate Antigen for the Initiation and Treatment of Multiple Sclerosis S19-071A Candidate Antigen for the Initiation and Treatment of Multiple Sclerosis
-
Bio-engineered mitochondria for targeted delivery to cells, tissue, and organs S24-026Bio-engineered mitochondria for targeted delivery to cells, tissue, and organs
-
STASH system: Enriching genetically modified cells with a single selectable surface marker S20-432STASH system: Enriching genetically modified cells with a single selectable surface marker