Docket #: S23-251
Activating Piezo1 channels for neuroprotection and axon regeneration in the retina and optic nerve
Glaucoma results from the loss of retinal ganglion cells (RGCs), the primary output neurons of the retina that connect to brain areas responsible for vision. Current treatments focus on lowering intraocular pressure but do not prevent blindness in about 25% of patients. Therefore, new strategies are needed to prevent RGC loss and regenerate their connections. For glaucoma and other optic neuropathies.
Stanford researchers have shown that the intravitreal delivery of small molecule Piezo1 agonists (like Yoda1, Jedi1/2) and the overexpression of Piezo1 via adeno-associated virus gene delivery, enhances RGC survival and axon protection. Piezo1 is a cation channel expressed in RGCs, with recent genetic analyses identifying Piezo1 variants that protect against glaucoma, suggesting it as a potential therapeutic target. In a mouse model of glaucoma, activating Piezo1 channels increased RGC and axon survival back to uninjured levels and restored visual function. This neuroprotective treatment can be used as an adjunct to current glaucoma treatments that lower intraocular pressure.
Figure:
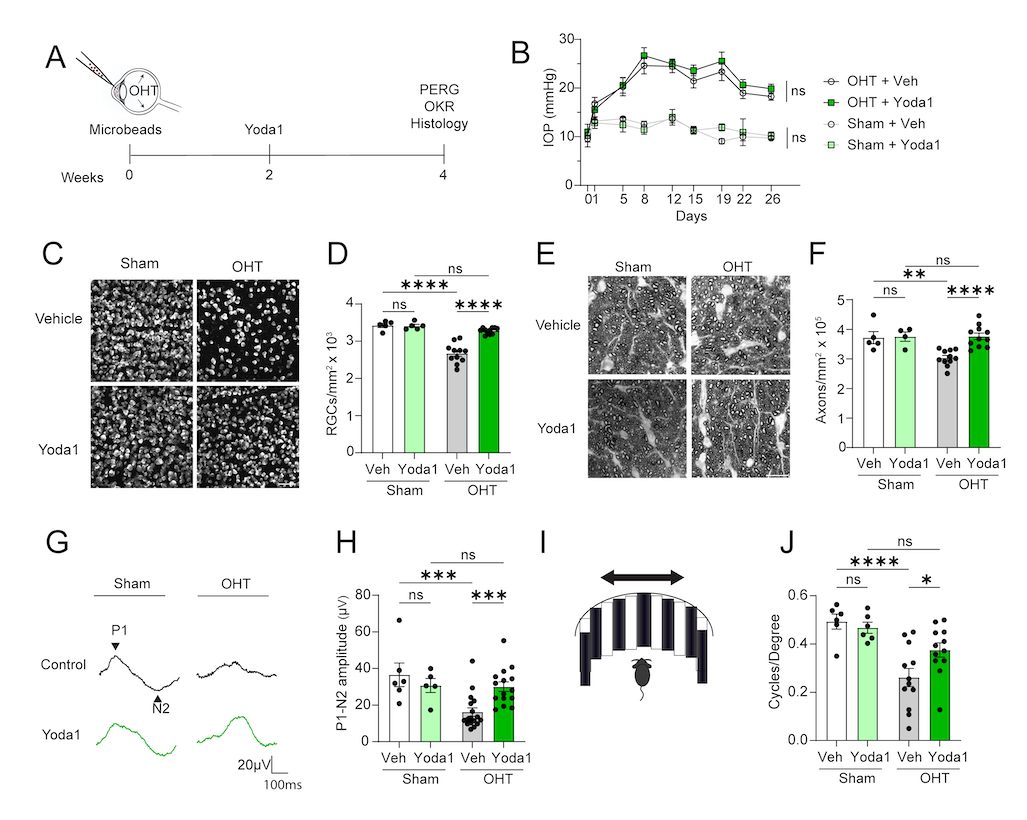
Piezo1 activation via its small molecule agonist Yoda1 promotes retinal ganglion cell neuroprotection and recovery of visual function.
(A) Schematic of experiment. Eyes were collected at 4 weeks for analysis.
(B) IOP after OHT induction or sham in Yoda1 and vehicle-treated eyes. (N = 5-11 per group, two-way ANOVA with repeated measures).
(C) Confocal images of RBPMS+ RGCs in whole-mount retinas treated with Yoda1 or vehicle 4 weeks after OHT induction or sham operation. Scale bar, 50 µm.
(D) Quantification of RGC survival.
(N = 5-11 per group, two-way ANOVA with Fisher's LSD).
(E) Light microscope images of semithin sections of optic nerve stained with p-phenylendiamine (PPD).
(F) Quantification of PPD staining. (N = 4-11 per group, two-way ANOVA with Fisher's LSD).
(G) Pattern electroretinogram (PERG) traces of eyes treated with Yoda1 or vehicle 4 weeks after OHT induction or sham operation.
(H) Quantification of P1-N2 PERG amplitude. (N = 5-17 per group, two-way ANOVA with Fisher's LSD).
(I) Schematic of optomotor response (OMR) test.
(J) Quantification of visual acuity in OMR represented as cycles/degree. (N = 6-12 per group, two-way ANOVA with Fisher's LSD).
All data are shown as mean ± SEM; * P0.05, **P0.01, ***P0.001, ***P00001; ns, not significant.
Stage of Development
Research - in vivo mouse models show neuroprotective effect and recovery of visual function.
Applications
- Glaucoma
- Ischemic optic neuropathy
- Optic neuritis
- Traumatic optic neuropathy
- Gene therapy
Advantages
- Robust neuroprotection in the eye
- Novel therapeutic target
- Novel mechanism of action
- Small molecule that can be formulated for easy delivery into the eye
- Animal models show improved visual performance
Related Links
Patents
- Published Application: WO2025019150
Similar Technologies
-
Neuroprotection and axon regeneration therapies for CNS axonopathies by modulating mitochondria trafficking S22-413Neuroprotection and axon regeneration therapies for CNS axonopathies by modulating mitochondria trafficking
-
Oligocarbonate molecular transporters across biological barriers S08-441Oligocarbonate molecular transporters across biological barriers
-
Chemically modified AAVs S21-280Chemically modified AAVs