Docket #: S17-075
Alloyed halide double perovskites as solar-cell absorbers
Solar cells containing halide perovskite absorbers have shown large improvements in power conversion efficiency over the last eight years and now exceed 20%. This makes them competitive with many commercial technologies like polycrystalline silicon and CdTe.
Stanford researchers at the Karunadasa Lab have previously proposed that double perovskites (e.g., Cs2AgBiBr6) could serve as less toxic substitutes for the lead perovskites (Stanford docket S15-455). However, all double perovskites known to date have large bandgaps (>1.95 eV), which are unsuitable for efficient sunlight absorption. This invention demonstrates that dilute alloying can dramatically decrease the bandgap of double perovskite absorbers made from nontoxic elements while maintaining the long carrier recombination lifetimes required for efficient charge extraction in a solar cell. This approach is general and can be applied to other dopants that can make a diverse family of double perovskites competitive with lead-based simple perovskite absorbers.
Figure
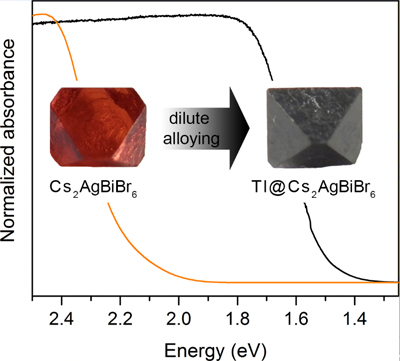
Figure description - Incorporation of dilute Tl impurities ( 1 atom%) results in a large bandgap reduction.This reduction is controlled by the amount of Tl incorporated and gives access to materials with bandgaps of ca. 1.4 eV, which is ideal for use in a single-junction perovskite solar cell. The carrier lifetime also remains long.
Stage of Research
Applications
- Use of dilute alloying/doping to decrease the bandgaps of double perovskites to the optimal range for single- junction and multi-junction solar cells
Advantages
- First demonstration of using alloying to modify the electronic structure in halide double perovskites
- Lower overall toxicity compared to (CH3NH3)PbX3
- More stable to moisture compared to (CH3NH3)PbI3
- More stable to heat compared to (CH3NH3)PbX3
- The alloyed double perovskite has long photoluminescence lifetimes (microsecond range at room temperature)
- The double perovskite structure allows for 1+, 2+, 3+, and 4+ metals while simple perovskites can only accommodate 2+ metals (e.g., Pb2+).
- General approach – can use other dopants that can engender similar effects in the broad family of halide double perovskites
Publications
- "Defect-induced band-edge reconstruction of a bismuth-halide double perovskite for visible-light absorption”, Slavney, A. H.; Leppert, L.; Bartesaghi, D.; Gold-Parker, A.; Toney, M. F.; Savenije, T. J.; Neaton, J. B.; Karunadasa, H. I. J. Am. Chem. Soc. 2017, 139, 5015.
- “A bismuth-halide double perovskite with long carrier recombination lifetime for photovoltaic applications”, Slavney, A. H.; Hu, T.; Lindenberg, A. M.; Karunadasa, H. I. J. Am. Chem. Soc. 2016, 138, 2138.
- U.S. Published Patent Application 20180277696, "ALLOYED HALIDE DOUBLE PEROVSKITES AS SOLAR-CELL ABSORBERS".
Patents
- Published Application: 20180277696
- Issued: 11,271,123 (USA)
Similar Technologies
-
Low-Temperature Synthesis of Polycrystalline Semiconductor Thin Films on Amorphous Substrates S10-129Low-Temperature Synthesis of Polycrystalline Semiconductor Thin Films on Amorphous Substrates
-
Ultrathin dielectric metasurface optical elements for easy fabrication and integration with semiconductor electronics S13-484Ultrathin dielectric metasurface optical elements for easy fabrication and integration with semiconductor electronics
-
Method of Organic Semiconductor Thin Film S12-369Method of Organic Semiconductor Thin Film